 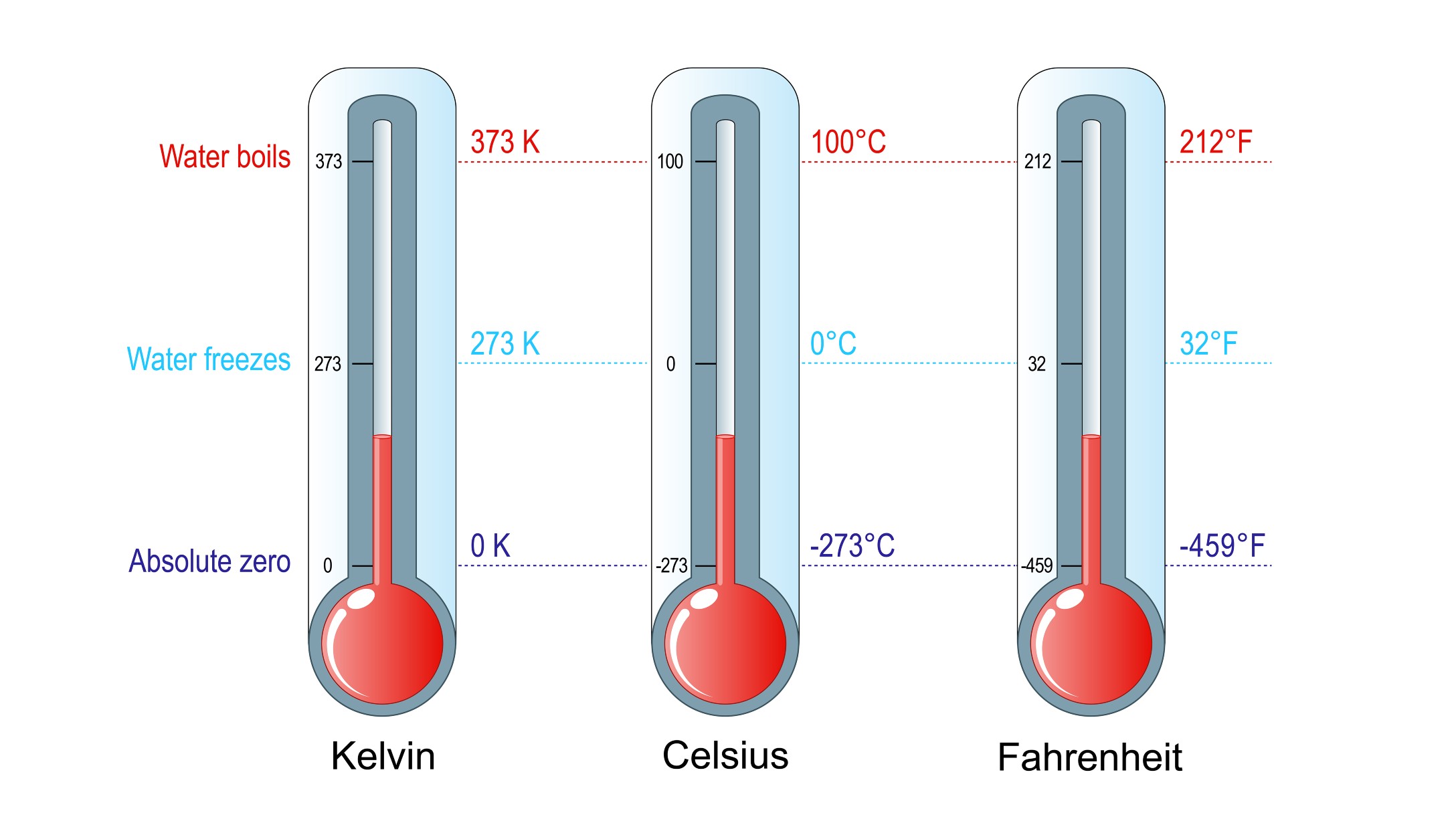 
The first room can be compared to a perfect crystal at absolute zero with low entropy, while the second room represents a system with higher entropy or disorder. Calculate the absolute third-law entropy of a 'solution' containing 1.08 moles of each gas. At 25☌ and 1 atm pressure, the absolute third. At 25☌ and 1 atm pressure, the absolute third-law entropies of methane and propane are 186.2 J K-1.mol1 and 1 respectively, in the gas phase. Calculate the absolute third-law entropy of a 'solution' containing 1.51 moles of each gas. where Q3 Q 3 is the amount of heat taken from the hotter reservoir and Q2 Q 2 is the. Question: At 25☌ and 1 atm pressure, the absolute third-law entropies of propane and methane are 269.9 J·K1·mol1 and 186.2 J·K1·mol1, respectively, in the gas phase. We have already seen that the efficiency of a Carnot cycle is given by. Now we can show this just from the laws of thermodynamics. The first room is meticulously organized, while the second room is in complete disarray. Although we have used the notion of absolute temperature, it was not proven. This order makes qualitative sense based on the kinds and extents of motion available to atoms and molecules in the three phases.A typical example to illustrate this concept could be considering two rooms. William Shiuk a year ago I wondered if some of these factors influencing entropy (moles, volume, state) are more dominant than others, e. 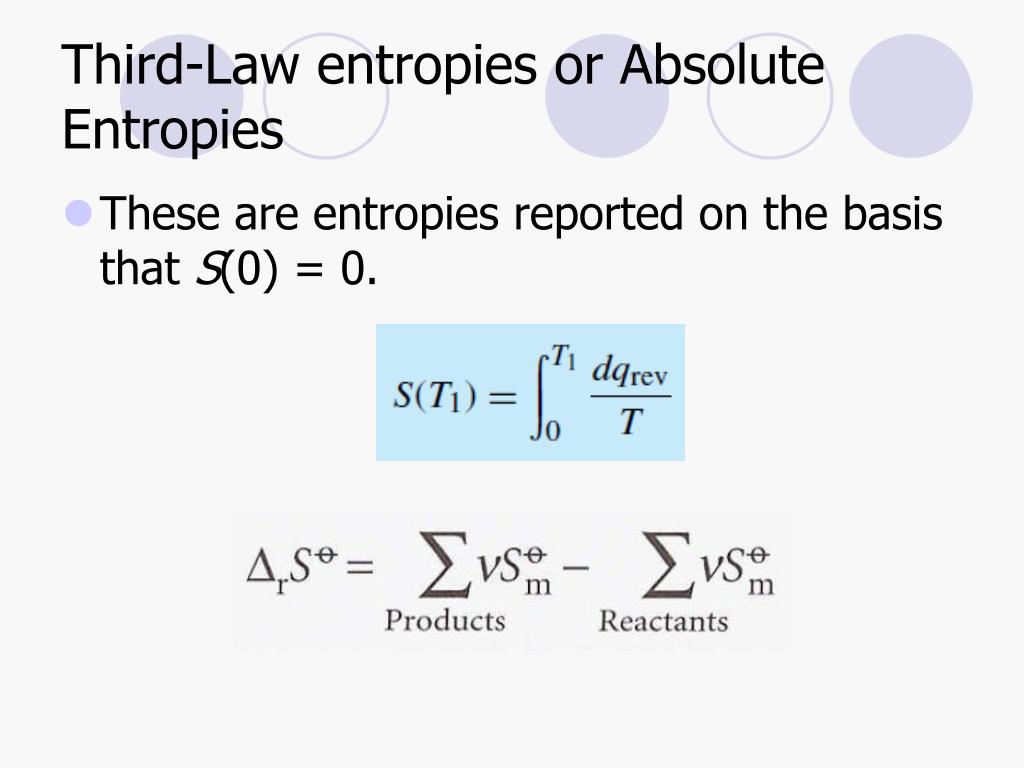
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
